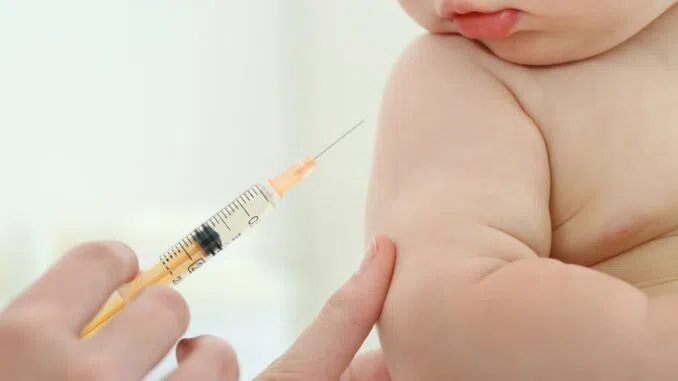
The UK government has approved the Pfizer-BioNTech Covid vaccine for children aged six months to four years old who have pre-existing health conditions. This means that around 60,000 young children in the UK are now eligible to receive two doses of the vaccine, with an interval of at least eight weeks between each jab.
Children with poorly controlled asthma, and heart, kidney, liver, or digestive system issues are among those who are eligible. The vaccine has been recommended as the best way to protect these vulnerable children from severe illness caused by the virus.
NHS sites will begin administering the jabs in mid-June, and parents are advised to wait for official communication before bringing their children forward. Children who have recently been infected with the virus should wait for at least four weeks before getting vaccinated.
According to the UK Health Security Agency, 51 children under the age of four have died from Covid since the start of the pandemic. However, this figure could be an overestimate, as it includes anyone who tested positive for the virus within four weeks of their death.
The Joint Committee on Vaccination and Immunisation (JCVI), which advises the UK government on the vaccine rollout, recommends that eligible children receive two doses of the Pfizer vaccine, with a low-dose formulation, and at least eight weeks apart. It further advises that third doses of the vaccine for immunosuppressed individuals will be issued in due course.
The JCVI’s recommendation comes after a review of Covid vaccine trials among children in the US, including safety data and monitoring the virus among youngsters in the UK. Data suggests that at-risk children aged six months to four years are seven times more likely to be admitted to intensive care with severe Covid.
Professor Wei Shen Lim, chair of the JCVI’s COVID-19 Committee, emphasizes that the vaccine is the best way to protect vulnerable children from a serious illness. Although Covid usually causes mild symptoms in most children, it can lead to severe illness in a small group of children with pre-existing health conditions. It is important to note that healthy children in the age group are not currently eligible for the vaccine.
Free Speech and Alternative Media are under attack by the Deep State. Real News Cast needs reader support to survive. Please Contribute via GoGetFunding